
Mifepristone. Using in Medical Abortion. Price
The most common questions from girls/women concerned about pregnancy safe early termination are: “What is the Mifepristone price in an online pharmacy?”. You will find the answer to these (and other) questions on this page.
You can check all prices on abortion pills in our online pharmacy. When you place an order for any full MTP kit, you will receive a complete package for early abortion and unlimited consultation with a professional gynecologist from start to finish the procedure!
Mifepristone. Pharmacology
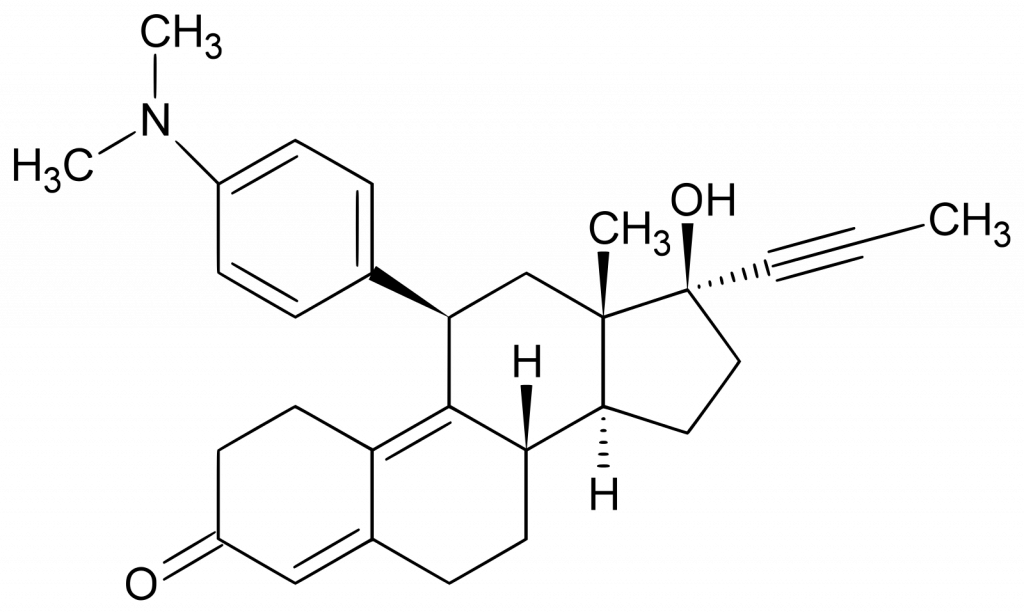
The Mifepristone pharmacological action is antiprogestogenic.
It blocks the action of progesterone at the receptor level. It has no gestagenic activity. Antagonism with glucocorticoids is noted at the competition expense at the receptor binding level.
Increases myometrial contractile activity by stimulating the interleukin release in uterine cells and increasing myometrial sensitivity to prostaglandin. Mifepristone is combined with a synthetic prostaglandin analog (e.g., Misoprostol) to intensify the MTP effects. Uterine contractions stimulation leads to the oviduct uterus and expulsion lining rejection.
Mifepristone for uterine leiomyoma may help to inhibit tumor growth and reduce the myomatous nodules and the uterus size.
Absolute bioavailability is 69%. When taken orally, Mifepristone 100-800 mg is rapidly absorbed in the gastrointestinal tract (GIT), with an average plasma concentration of 2.5 mg/l after 0.7-1.5 hours. In plasma, Mifepristone 98% is in a protein-bound state. After 12-72 hours, the plasma concentrations are reduced by half. It is important to note that there is no difference in blood concentrations between 72 hours after Mifepristone 100, 200, 400, 600, or 800 mg administration.
Abortion Pill Kit
Buy Abortion Pill Kit online in the US, Europe, and Worldwide
150$
Add to cart
AntiPREG Kit
Buy Abortion Pills online in the US, Europe, Worldwide
150$
Add to cart
Mifepristone Misoprostol combipack
Buy Mifepristone Misoprostol combipack online in the US, Europe, Worldwide
150$
Add to cart
Cytolog
Buy Cytolog online in the US, Europe, and Worldwide
150$
Add to cart
Cytotec
Buy Cytotec online in the US, Europe, and Worldwide
150$
Add to cart
Generic RU-486
Buy Generic RU-486 online in the US, Europe, Worldwide
150$
Add to cart
Mifeprex
Buy Mifeprex online in the US, Europe, and Worldwide
150$
Add to cart
Mifepristone
Buy Mifepristone online in the US, Europe, and Worldwide
150$
Add to cart
Misoprostol
Buy Misoprostol online in the US, Europe, and Worldwide
150$
Add to cart
Misoprost-200
Buy Misoprost-200 online in the US, Europe, Worldwide
150$
Add to cart
CONTRAKIT
Buy MTP Kit online in the US, Europe, Worldwide
150$
Add to cart
KillPreg Kit
Buy MTP kit online in the US, Europe, Worldwide
150$
Add to cart
Mifepristone + Misoprostol Kit
Buy Mifepristone + Misoprostol Kit online in the US, Europe, and Worldwide
150$
Add to cart
MTP Kit
Buy MTP Kit online in the US, Europe, Worldwide
150$
Add to cartIndications for Mifepristone

- Early uterine pregnancies medical interruption (up to amenorrhoea 77 days – 11 weeks’ gestation), confirmed by ultrasound in combination with Misoprostol.
- Labour preparation and induction in a term pregnancy.
- Emergency (postcoital) contraception within unprotected intercourse 72 hours or if the method used cannot be considered reliable (10 mg tablets).
- Uterine leiomyoma treatment (up to 12 weeks gestation) (50 mg tablets).
Contraindications for Mifepristone

General contraindications
- Hypersensitivity to the active substance;
- Adrenal insufficiency and long-term treatment with glucocorticoids;
- Acute or chronic renal and hepatic insufficiency;
- Porphyria
For pregnancy termination
- The a uterine scar presence (relative contraindication);
- The female genitalia Inflammatory diseases;
- The severe extragenital pathology presence;
- Haemostasis disorders (including previous treatment with anticoagulants);
- Anaemia (relative contraindication).
- Do not use in women over 35 age years who smoke (without prior consultation with a general practitioner);
- Suspected ectopic pregnancy;
- Pregnancy not clinically confirmed;
- More than 77 days Pregnancy from the last menstrual period first day;
- Pregnancy resulting from the intrauterine devices use;
- Pregnancy after hormonal contraception withdrawal;
- Uterine myoma (relative contraindication).
For labor preparation and induction
- Severe gestosis;
- Pre-eclampsia;
- Eclampsia;
- Premature pregnancy;
- Preterm pregnancy;
- Myoma uteri.
For the uterine leiomyoma treatment
- Leiomyoma exceeding 12 weeks’ gestation;
- Ovarian tumors and/or endometrial hyperplasia;
- Myomatous nodules Submucosal location;
- Pregnancy;
- Lactation period.
Limitations to use
Use with caution in the following patients:
- With bronchial asthma;
- With chronic obstructive pulmonary disease;
- With cardiovascular diseases (including arterial hypertension, heart rhythm disturbances, chronic heart failure) or predisposition to them.
Mifepristone Side effects

Gastrointestinal effects:
- Discomfort is a feeling in the lower abdomen;
- Nausea;
- Vomiting;
- Diarrhoea.
General symptoms:
- Weakness;
- Headache;
- Dizziness;
- Hyperthermia;
- Rashes on the skin.
Related to the pregnancy termination procedure (optional):
- Heavy bleeding;
- Pain in the lower abdomen;
- Inflammation of the uterus and appendages;
- Exacerbation of uterine and urinary tract infections.
Against the combined treatment background with Misoprostol (additional):
- Dyspepsia;
- Insomnia;
- Asthenia;
- Pain in the leg;
- Restlessness;
- Anaemia, decrease in hemoglobin (not more than 2 g/dl);
- Fainting spells;
- Leucorrhea.
In emergency contraception case (optional):
- Bleeding from the genital tract;
- Menstrual disorders.
In the leiomyoma treatment (additional):
- Menstrual disorders;
- Amenorrhea.
Interactions with other drugs

Avoid use with non-steroidal anti-inflammatory drugs (NSAIDs) (including acetylsalicylic acid) because of possible effectiveness changes.
Overdose
No adverse reactions occur after Mifepristone administration up to 2000 mg a dose. In overdose cases, adrenal insufficiency may occur. The therapy is symptomatic.
Mifepristone dosage and administration
For pregnancy termination (in combination): 200 mg once, followed by Misoprostol. A repeat ultrasound check-up is necessary after the bleeding onset, and a clinical examination is required after 10-14 days (at the earliest). If there is no effect on the 14th day (incomplete abortion, ongoing pregnancy), vacuum aspiration is followed by the aspirate histological examination. Please refer to the Instructions for Use for further details.
For preparation and labor induction: 200 mg once (in a doctor’s presence). After 24 hours, 200 mg repeated intake. After 48-72 hours, the birth canal state is assessed, and, if necessary, prostaglandin (Misoprostol) or Oxytocin is prescribed.
For contraceptive purposes: for 72 hours after unprotected intercourse, Mifepristone (10 mg) 1 tablet in the menstrual cycle any phase. To maintain the contraceptive effect, it is necessary to refrain from eating 2 hours before and 2 hours after the drug administration.
Uterine leiomyoma treatment: 50 mg once daily; the treatment course is three months under the attending gynecologist’s supervision.
Precautions
Massive uterine bleeding may require immediate surgical intervention (curettage).
The woman must be informed that the pregnancy a surgical termination will be necessary if Mifepristone treatment fails (possible congenital disabilities due to its effect on the fetus).
Its use requires general measures accompanying abortion, including rhesus-alloimmunization prevention.
Breast-feeding should be discontinued for seven days after the drug application (if 200 mg a dose has been used).
Effect on the ability to drive
Mifepristone may cause dizziness, and caution should be exercised when driving motor vehicles and operating complex machinery.
Patients with artificial heart valves or infective endocarditis should be treated prophylactically with antibiotics during Mifepristone administration.
Mifepristone (10 mg tablets) is not recommended for regular use as planned permanent contraception; it does not protect against sexually transmitted diseases and HIV.
Conditions for dispensing from pharmacies
The drug may be supplied only to obstetrical and gynecological clinics belonging to the public health system and municipal and privately-owned institutions with a license for activity of this type.
Storage conditions and terms
The drug should be stored in a dry place, protected from light, out of children’s reach: shelf life – 2 years. Please do not use it after the expiry date stated on the package.
The page shows the Mifepristone tablet’s price and costs at our online pharmacy Your-Safe-Abortion.com. And also the drug Mifepristone description, its mechanism of action, the effects (including side effects) in medical abortion (MTP). The dose required for a safe MTP is given.

 Français
Français