Product description
MTP Kit — The Complete Product Guide
Facing an unplanned pregnancy can be an emotionally overwhelming and challenging experience. In such situations, considering options like abortion becomes crucial for some women. The abortion pill kit, also known as a medical abortion or medication abortion, has gained popularity as a safe and effective method to terminate an early pregnancy. This article aims to provide you with a comprehensive guide on the MTP Kit, its components, usage, precautions, and important considerations.
Our online pharmacy has other abortion pill kits in stock that contain identical doses of Mifepristone and Misoprostol: Combipack Mifepristone + Misoprostol, Abortion Pills Kit, and Mifepristone + Misoprostol Kit.
Understanding the MTP Kit
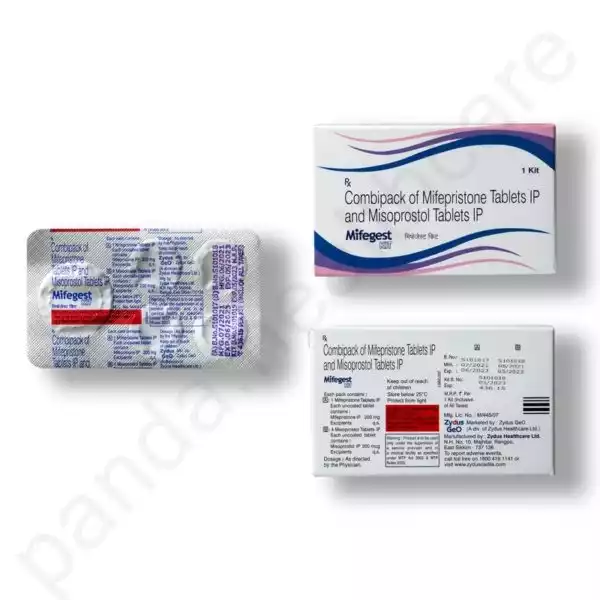
An MTP Kit is typically a combination of two medications, Mifepristone and Misoprostol. Mifepristone is often referred to as the initial pill, while Misoprostol is the follow-up medication. These medications work together to induce a miscarriage-like effect and terminate an early pregnancy.
1. Mifepristone:
Mifepristone is taken first, usually under medical supervision. It works by blocking the hormone progesterone, which is essential for maintaining pregnancy. By inhibiting progesterone, it causes the lining of the uterus to break down, preventing the pregnancy from progressing.
2. Misoprostol:
Misoprostol is taken a day or two after Mifepristone. For more details, see Instruction. Its primary function is to cause contractions in the uterus, leading to the expulsion of the pregnancy. This medication is usually taken at home, allowing the individual to have privacy and control over their environment during the process.
Guidelines for Using the MTP Kit:
It is important to remember that while the abortion pill kit is considered safe, it should only be used under the guidance and supervision of a healthcare professional. Here are some general guidelines regarding the usage of the MTP Kit:
- Consult a healthcare professional: Before considering a medical abortion, it is essential to consult a healthcare professional who can evaluate your specific circumstances and ensure it is a suitable option for you.
- Timing: The abortion pill kit is most effective when used within the first 10 weeks of pregnancy. It is crucial to follow the recommended dosage and timing instructions provided by your healthcare provider.
- Possible side effects: The process of medical abortion can cause side effects such as cramping, bleeding, nausea, and fatigue. These symptoms generally subside within a few days. However, it is important to seek medical attention if you experience severe or prolonged symptoms.
- Follow-up appointment: A follow-up appointment with your healthcare provider is essential to ensure the successful completion of the abortion and monitor any potential complications.
Precautions and Important Considerations:
While the MTP Kit can be an option for terminating an early pregnancy, certain precautions and considerations should be kept in mind:
- Ectopic pregnancy: The abortion pill kit is not effective for terminating ectopic pregnancies, which occur when the fertilized egg implants outside the uterus. Ectopic pregnancies require immediate medical attention, as they can be life-threatening.
- Emotional support: Going through an abortion can be emotionally challenging for some individuals. It can be beneficial to seek support from trusted friends, family, or professional counselors during this time.
- Confidentiality: It is crucial to ensure your privacy and confidentiality throughout the process. Make sure to follow the instructions provided to dispose of the used medications safely.
Consulting a Healthcare Professional:
While this article aims to provide general information on abortion pill kits, it is important to remember that each individual’s circumstances are unique. Consultation with a healthcare professional is critical to evaluate your specific situation, as they can provide personalized guidance, address concerns, and offer appropriate medical advice.

 Français
Français Deutsch
Deutsch Español
Español Русский
Русский
The information was protected by a bar code on the delivered package; and the goods was housed in a suitable box. The items were bubble-wrapped inside.